Molecular Pharming Solution assists clients in efficiently launching their products into the market

End-to-End CDMO & Integrated Services
for Plant-Based Recombinant Proteins
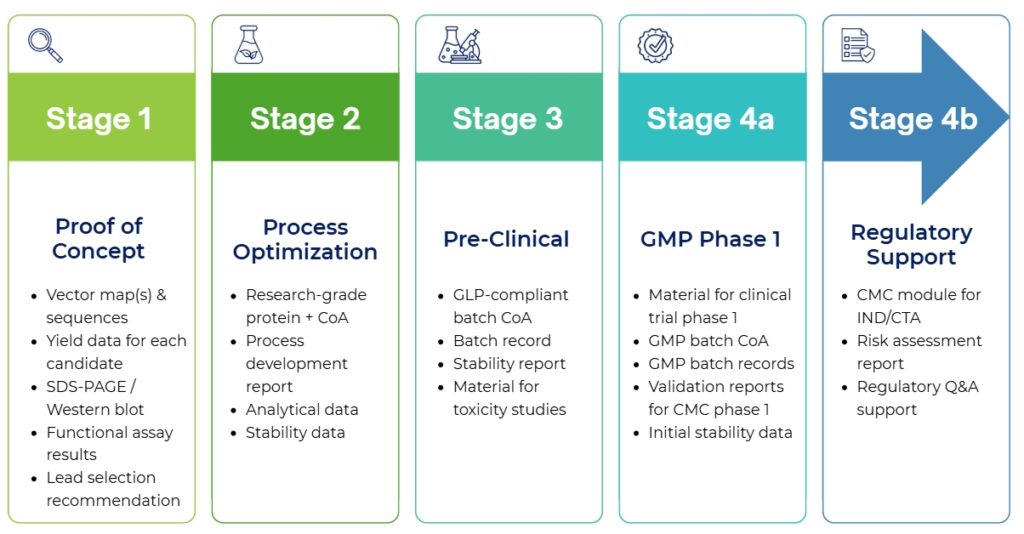
From construct design to purified protein, BaiyaPharming™ delivers
integrated CDMO services using a plant-based transient expression
platform in Nicotiana benthamiana. Our animal-free workflow enables
fast, flexible, and scalable protein production
for research and early-stage development, with built-in quality control and regulatory readiness.
for research and early-stage development, with built-in quality control and regulatory readiness.
View Service Capabilities